Sodium bromide lattice energy equation8/15/2023 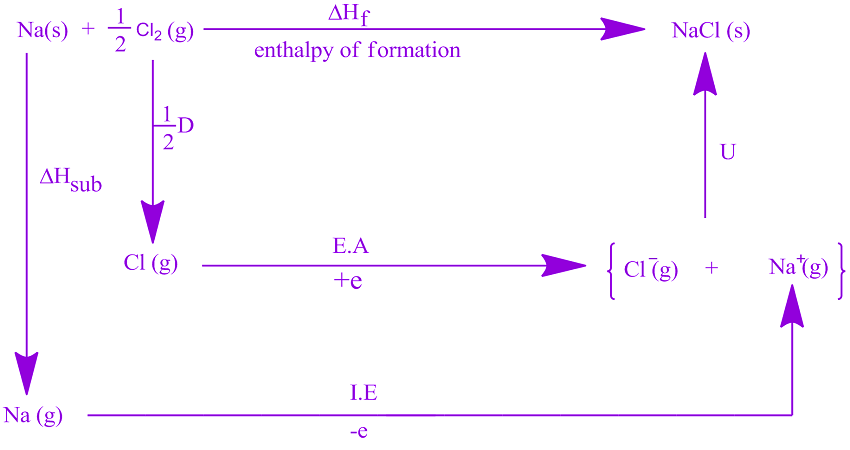
Draw and label a BornHaber cycle to calculate the lattice energy of magnesium bromide. If you get a different value, please let me know. The lattice energy of magnesium bromide, MgBr 2, can be calculated using the enthalpy changes shown in the table. Given the lattice energy of NaBr 736 kJ/mol, the ionization energy of Na 496 kJ/mol and the electron affinity of Br 325 kJ/mol, calculate the H o. Answerĭiscussion: This number has not been checked. Lattice energy can be a very complicated process but is often simplified by using Coulomb’s law. The attraction of the two ions releases energy and the process is exothermic. Evaluate the energy of crystallization, E cryst. Lattice energy refers to the energy that is released when two oppositely charged gaseous ions attract to each other and form an ionic solid. 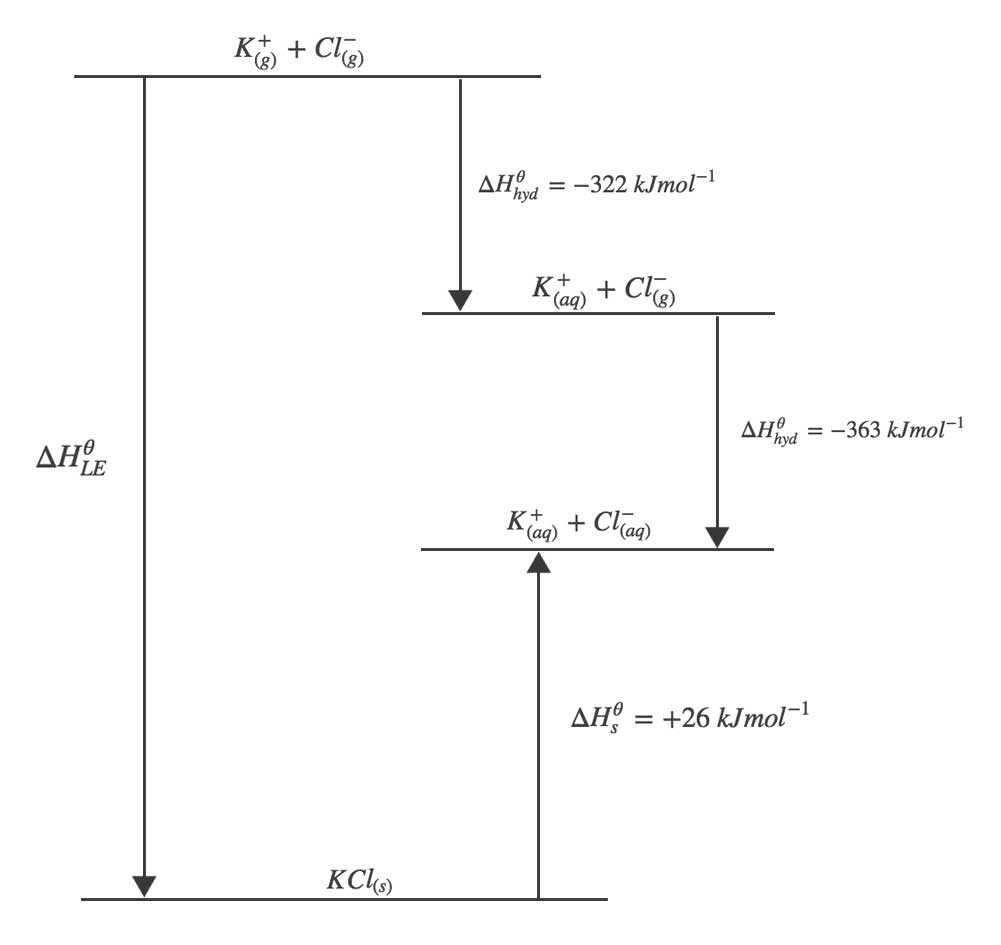
Skill: Evaluate the lattice energy and know what values are needed.Īssume the interionic distance for NaCl 2 to be the same as those of NaCl ( r = 282 pm), and assume the structure to be of the fluorite type ( M = 2.512). AnswerĮnergy of crystallization is -3527 kJ/mol Evaluate the energy of crystallization, E cryst for CaO. Lime, CaO, is know to have the same structure as NaCl and the edge length of the unit cell for CaO is 481 pm. 5 9701/4 O/N/02 Turn over (c) Use your cycle, the following data, and further data from the Data Booklet to calculate a value for the lattice energy of sodium oxide. Which would have more negative lattice energy, lithium chloride or lithium bromide MgO is almost four times greater then the lattice energy of NaF, why V. Which one of the following has the largest lattice energy? LiCl, NaCl, CaCl 2, Al 2O 3 AnswerĬorrundum Al 2O 3 has some covalent character in the solid as well as the higher charge of the ions. Formula: NaBr Hill system formula: Br 1 Na 1 CAS registry number: Formula weight: 102. Skill: Explain the trend of lattice energy. Which one of the following has the largest lattice energy? LiF, NaF, CaF 2, AlF 3 Answer When methods to evaluate the energy of crystallization or lattice energy lead to reliable values, these values can be used in the Born-Haber cycle to evaluate other chemical properties, for example the electron affinity, which is really difficult to determine directly by experiment. 
Much more should be considered in order to evaluate the lattice energy accurately, but the above calculation leads you to a good start. In addition to presenting comparative calculations by two approaches for the total lattice potential energy of sodium bromide dihydrate, NaBr 2H 2 O, found to take the value 803.9 kJ mol -1, we investigate the influence of the size and nature of the basis set used to generate multipole moments in a Hartee-Fock calculation which are. So I am going to rewrite it as a table.\( \newcommand\] The equation is: Lattice enthalpy Heat of formation- Heat of sublimation- (1/2 Dissociation energy. I have drawn this cycle very roughly to scale, but that is going to become more and more difficult as we look at the other two possible formulae. The lattice formula, or the Born-Haber cycle equation, is derived from the Born-Haber cycle. The larger value of Q 1 × Q 2 for the sodium ionoxide ion interaction means it will release more energy. So the compound MgCl is definitely energetically more stable than its elements. For the interaction of a sodium ion with an oxide ion, Q 1 1 and Q 2 2, whereas for the interaction of a sodium ion with a bromide ion, Q 1 1 and Q 2 1. \( \newcommand\]įind two routes around this without going against the flow of any arrows.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
