Element definition and example8/15/2023 Other compounds are made from different combinations of atoms, like water… carbon dioxide… and table salt.Īnd that’s it! Nearly everything in this room, in your room, and in the entire universe, is made of: elements with one kind of atom, compounds containing different types of atoms chemically bonded together and mixtures of different elements and compounds together. Iron sulfide, a new substance, has been formed because the iron and sulfur atoms are now chemically bonded together. 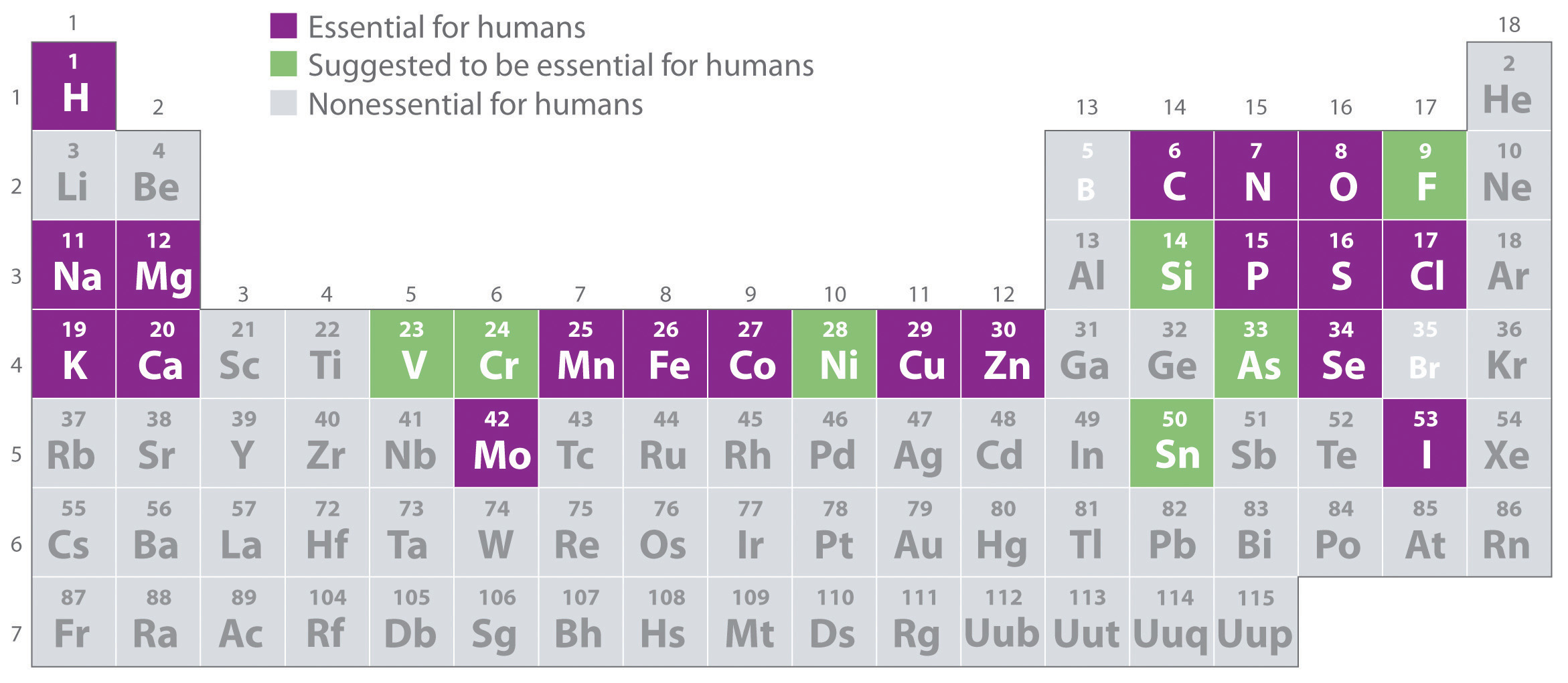
The iron can no longer be removed by a magnet because it’s bonded to the sulfur. Examples of compounds include table salt or sodium chloride (NaCl, an ionic compound), sucrose (a molecule), nitrogen gas (N 2, a covalent molecule), a sample of copper (intermetallic), and water (H 2 O, a covalent molecule). atoms of the two elements are now joined together by chemical bonds. When the iron and sulfur are heated together… We can still separate the elements in the mixture. The different elements are not joined together. This is now a mixture of the elements iron and sulfur. When we mix two different pure substances together, like this, it’s a mixture. It contains only sulfur atoms, and nothing else, so it is pure. It is made of only one type of atom: iron atoms. Some substances, like particles of this iron, contain only one kind of atom. E.g petrol in water, sand in water.Everything we can see and touch, and quite a lot that we can’t as well, is made of tiny particles called atoms. The type of mixture in which the composition is not uniform is known as a heterogeneous mixture. A homogeneous mixture exists in only one phase. The type of mixture in which the composition is uniform throughout the mixture. Heterogeneous mixture Homogeneous mixture Essential element in biological science is the chemical elements that have biological functions to sustain the life of living organisms such as plants or. The major component in a solution that dissolves the solute is known as a solvent. The minor component in a solution that is dissolved in the solvent is known as solute. Components of a mixtureĪ mixture is composed of two components namely The constituents of a mixture can be separated through various physical methods e.g Distillation, Filtration, and Crystallization. It is an impure substance.Ī mixture is obtained when two or more elements or compounds are mixed in any ratio such that their constituents retain their original properties. Compound ExamplesĪ physical combination of two or more substances in any ratio is known as a mixture. The formula shows the elements which chemically combined to form that particular compound. The constituents of a compound are represented through a chemical formula. Changing this ratio will yield an entirely different compound. A molecule is a substance that is made out of two or more atoms bonded via chemical bonds. 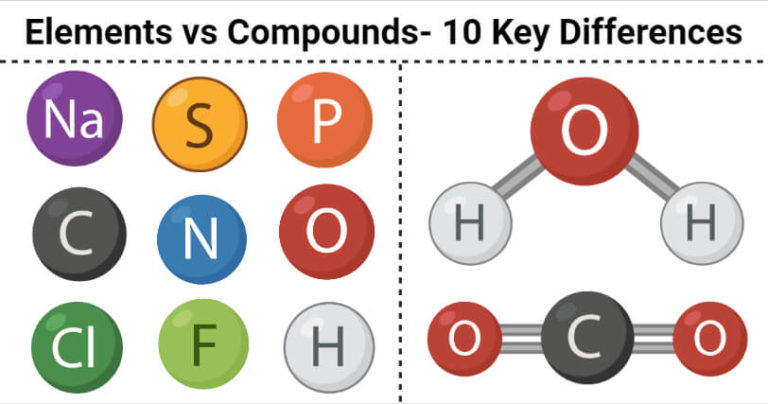
In hydrogen peroxide, the ratio of hydrogen to oxygen is always 2:2. An element is a substance that cannot be broken down further by chemical means. Which is applied to the skin as an antiseptic to prevent infection from minor cuts? Hydrogen Peroxide is an example of a compound. The most essential property of a compound is its constant mass ratio. a small amount of an emotion or quality: There was certainly an element of truth in what she said. A data element is defined by size (in characters). As a compound is a pure substance, its constituents cannot be separated through physical means. Any unit of data defined for processing is a data element for example, ACCOUNT NUMBER, NAME, ADDRESS and CITY. ExplanationĬompounds are formed by the chemical combination of elements whose properties are entirely distinct from those of their parent elements. 
A pure substance that is formed when two or more elements combine chemically in a definite proportion by mass is known as a compound.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
